A Florida State University chemist has developed a method to rapidly assemble significantly complex natural molecules with potential in biomedical applications, opening the door for novel drug therapies based on the molecule’s structure.
James Frederich, the Werner Herz Associate Professor of Chemistry and Biochemistry, and his team are the first to fully synthesize fusicoccadiene, a precursor to an emerging treatment in cancer chemotherapy. Their work was recently published in the Journal of the American Chemical Society.
“The Frederich Laboratory specializes in the synthesis of architecturally complex natural products that we believe have special translational potential, especially in medicine, but are currently inaccessible in a practical manner,” Frederich said. “We build complex structures from scratch by extending existing chemical methods and developing entirely new ones.”
The functions of biomolecules, or the critical roles that proteins, lipids and other molecules serve in driving life-sustaining cellular processes, are directly determined by their structure. By studying architecturally complex substances from the natural world — like fusicoccum amygdali, the fungus that produces fusicoccadiene — scientists can alter molecules and then translate those chemical structures into starting points for drug discovery.
What it is
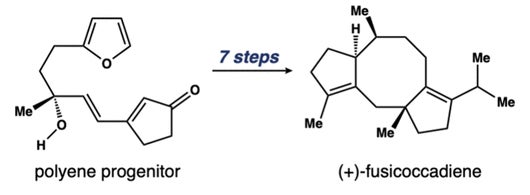
Fusicoccadiene is the hydrocarbon precursor molecule to fusicoccanes, a family of natural molecules stemming from fungi that hold significant potential in biomedical applications. Several fusicoccanes, including fusicoccin A and cotylenin A, can induce cell death in cancer cells by sensitizing them to intrinsic cell death mechanisms.
While the structure of fusicoccadiene — a 5-8-5 system of rings — is critical in the translation to novel drug therapies, it’s also extraordinarily complex and difficult to synthesize in labs. This system comprises two 5-membered rings fused to a central 8-membered ring that makes up the molecule’s core.
“Realizing the synthetic blueprint to prepare fusicoccadiene was very challenging,” Frederich said. “This synthesis is the culmination of several doctoral thesis projects spanning almost a decade. Chemical synthesis requires great resolve from both students and principal investigators, and it requires a special creativity.”
How it’s made
The synthetic technique used in the Frederich Lab to prepare fusicoccadiene includes converting one compound of a polyene progenitor into a different compound using light to facilitate the chemical processes. After the molecule is produced, researchers perform modifications that allow them to alter the molecular structure at precise, site-specific locations to yield desired compounds and specific spatial arrangements, producing different functionalities and applications.
“Instead of designing a molecule for target-specific endpoints, we envisioned an assembly scheme that could capture new, non-natural compositions of matter for future iterations of the molecule that can be used in medicine,” Frederich said. “Our approach focuses on direct construction of the 5-8-5 nucleus in the early stages of molecular formation. We then leverage a range of certain reactions to decorate the periphery of the structure with a range of functionality.”